|
1/14/2024 0 Comments Reactivity of dienophiles 
17 Regarding the former, this is illustrated by the difference in the. Which dienes react more rapidly than A Enter the letters corresponding to your selections in the answer box. Using 1,3-butadiene ( A) as a reference, consider the relative reactivity of each in a Diels-Alder reaction with the dienophile maleic anhydride 1. Traceless Thioacid-Mediated Radical Cyclization of 1,6-Dienes. Diene Reactivity in the Diels-Alder Reaction Examine the diene formulas A through G. The dienophile \textit increasing reactivity is shown in the picture below. Illustrating the strategic value of this method for synthetic planning, the reaction of 4- and 2-vinylpyridines with a variety of benchmark dienes follows predictable trends of reactivity and regioselectivity commonly observed with conventional dienophiles (Fig. Cyclopropenes are small and highly reactive compounds however, their synthesis poses. The initial bonding interaction reflects this electron imbalance, with the two new sigma-bonds being formed simultaneously, but not necessarily at equal rates.Let's take a look at the structures of the given compounds in order to determine their reactivity as dienophiles in Diels-Alder reaction. Introduction The acceleration of Diels-Alder reactions by Lewis acid catalysts has had a tremendous impact in organic synthesis since its discovery by Yates in 1961.1 By coordinating with carbonyl-conjugated dienophiles, Lewis acids affect the reactivity and the diastereoselectivky outcome of the reaction. In its usual form, the diene component is electron rich, and the best dienophiles are electron poor due to electron withdrawing substituents such as CN, C=O & NO 2. For many acyclic dienes the s-trans conformer is more stable than the s-cis conformer (due to steric crowding of the end groups), but the two are generally in rapid equilibrium, permitting the use of all but the most hindered dienes as reactants in Diels-Alder reactions. The Diels-Alder reaction is a single step process, so the diene component must adopt an s-cisconformation in order for the end carbon atoms (#1 & #4) to bond simultaneously to the dienophile. Toward 3,6-dipyridyl-s-tetrazine in MeOH at 25 C, the strained derivative is 19 and 27 times more reactive than the parent trans-cyclooctene and 4E-cyclooct-4. It is the prototypical example of a pericyclic reaction. The Diels-Alder cycloaddition is classified as a process because the diene has four pi-electrons that shift position in the reaction and the dienophile has two. In organic chemistry, the DielsAlder reaction is a chemical reaction between a conjugated diene and a substituted alkene, commonly termed the dienophile, to form a substituted cyclohexene derivative. 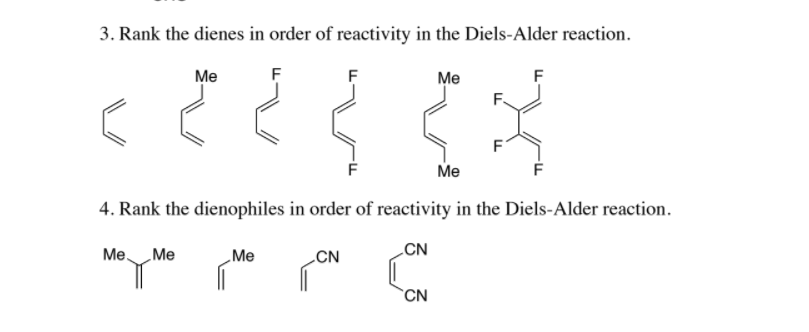
The chiral group in the template is labelled with. Cycloaddition reactions studied in this work between a chiral anthracene template ANT and eleven dienophiles 1 A to 3 C. The reactants used in such reactions are a conjugated diene, simply referred to as the diene, and a double or triple bond coreactant called the dienophile, because it combines with (has an affinity for) the diene. Chiral anthracene templates and dienophiles used for DA cyclo-additions. The Diels-Alder reaction is an important and widely used method for making six-membered rings, as shown on the right. The cycloaddition converts these pi-bonds into new sigma-bonds (colored green), and this transformation is then designated a cycloaddition, to enumerate the reactant pi-electrons that change their bonding location. In the hypothetical ethylene dimerization on the left, each reactant molecule has a pi-bond (colored orange) occupied by two electrons. The following diagram illustrates two cycloadditions, and introduces several terms that are useful in discussing reactions of this kind. 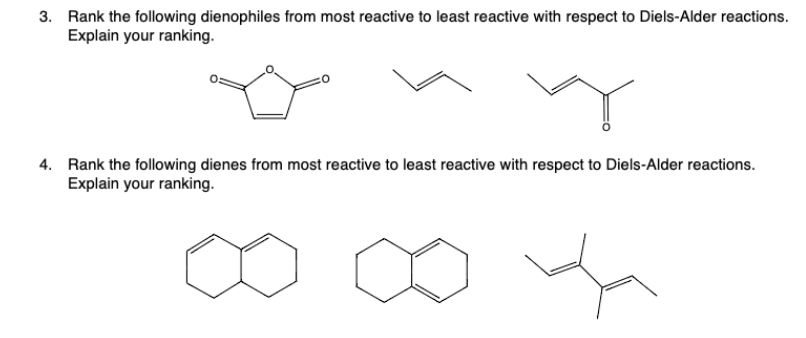
SN1 and SN2 reactions - kinetics, order of reactivity of alkyl halides. This does not occur under normal conditions, but the cycloaddition of 1,3-butadiene to cyanoethene (acrylonitrile) does, and this is an example of the Diels-Alder reaction. Stability of conjugated dienes, Diel-Alder, electrophilic addition, free radical. Cyclopentadiene and cyclopropene are unusual, in that they exhibit rapid Diels-Alder reactivity despite their lack of activating electron withdrawing or. When this occurs, two pi-bonds are converted to two sigma-bonds, the simplest example being the hypothetical combination of two ethene molecules to give cyclobutane. Reactivity increase of dienophiles by change of the ester substituents into the acid chlorides: cycloaddition reaction of the diacid chloride of 3,10-hexacyclo 10.2.1.15,8.02,11.03,10. A cycloaddition reaction is the concerted bonding together of two independent pi-electron systems to form a new ring of atoms. The unique character of conjugated dienes manifests itself dramatically in the Diels-Alder Cycloaddition Reaction. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
